Abstract
Research Article
Insilico investigation of TNFSF10 signaling cascade in ovarian serous cystadenocarcinoma
Asima Tayyeb* and Zafar Abbas Shah
Published: 02 July, 2019 | Volume 3 - Issue 1 | Pages: 025-034
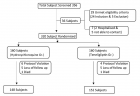
The ovarian serous Cystadenocarcinoma shared large number of deaths in gynecologic carcinoma. It has various numbers of molecular events from initiation to progression and at advance stage, surgery is the end product of such molecular signaling. We assess in this study the whole mechanistic view of TNFSF10 network which has the ideal apoptotic causing identity. We used fresh insilico strategy to uncover the secrets and inter-links from its protein-protein interaction complex. We retrieved the TNFSF10 signaling network from STRING database (www.string-db.org). The network contains 25 nodes and 152 edges with clustering presentation. After retrieval, we performed gene enrichment and characterization analysis of network from WebGestalt toolkit (www.webgestalt.com). Finally, we examined the participation of whole network in ovarian cancer progression from cBioPortal, a cancer genomic data portal (www.cbioportal.org). Our results showed that majority of cases have loss of function of death receptors (DR4 and DR5) that are the main unit of initiation of apoptotic signaling. Most of downstream signaling members showed amplification that regulates cell proliferative pathways including NFkB pathway. TNFSF10 cluster has loss of function and in future it gain attention for further research studies to discover its interactome level view for valuable therapy. FAS cluster has large number of members and majority showed amplification rendering them as co-targets for combinational drug designing.
Read Full Article HTML DOI: 10.29328/journal.acst.1001005 Cite this Article Read Full Article PDF
Keywords:
Ovarian cancer; TNFSF10; TNFSF10; FAS
References
- Pitti RM, Marsters SA, Ruppert S, Donahue CJ, Moore A, et al. Induction of apoptosis by Apo-2 ligand, a new member of the tumor necrosis factor cytokine family. J Biol Chem. 1996; 271:12687-12690. Ref.: http://bit.ly/2Nso1Su
- Wiley SR, Schooley K, Smolak PJ, Din WS, Huang CP, et al. Identification and characterization of a new member of the TNF family that induces apoptosis. Immunity. 1995; 3: 673-682. Ref.: http://bit.ly/2LxPDmv
- Kimberley FC, Screaton GR. Following a TRAIL: update on a ligand and its five receptors. Cell Res. 2004; 14: 359. Ref.: http://bit.ly/327eofe
- Diehl GE, Yue HH, Hsieh K, Kuang AA, Ho M, et al. TRAIL-R as a negative regulator of innate immune cell responses. Immunity. 2004; 21: 877-889. Ref.: http://bit.ly/2LulNzj
- Cretney E, Takeda K, Yagita H, Glaccum M, Peschon JJ, et al. Increased susceptibility to tumor initiation and metastasis in TNF-related apoptosis-inducing ligand-deficient mice. J Immunol. 2002; 168: 1356-1361. Ref.: http://bit.ly/2xnjPZf
- Abdulghani J, El-Deiry WS. TRAIL receptor signaling and therapeutics. Expert Opin Ther Targets. 2010; 14: 1091-1108. Ref.: http://bit.ly/2JpayFw
- Wang X. The expanding role of mitochondria in apoptosis. Genes Dev. 2001; 15: 2922-2933. Ref.: http://bit.ly/2FM402U
- Lin Y, Devin A, Cook A, Keane MM, Kelliher M, et al. The death domain kinase RIP is essential for TRAIL (Apo2L)-induced activation of IκB kinase and c-Jun N-terminal kinase. Mol Cell Biol. 2000; 20: 6638-6645. Ref.: http://bit.ly/2XFqZH3
- Wajant H. TRAIL and NFκB signaling—a complex relationship. Vitamins & Hormones. 2004; 67: 101-132. Ref.: http://bit.ly/2YptNoF
- Wajant H, Gerspach J, Pfizenmaier K. Tumor therapeutics by design: targeting and activation of death receptors. Cytokine & growth factor reviews. 2005; 16: 55-76. Ref.: http://bit.ly/2ROkbS2
- Wang X, Chen W, Zeng W, Bai L, Tesfaigzi Y, et al. Akt-mediated eminent expression of c-FLIP and Mcl-1 confers acquired resistance to TRAIL-induced cytotoxicity to lung cancer cells. Mol Cancer Ther. 2008; 7: 1156-1163. Ref.: http://bit.ly/304lz5Y
- Chen W, Bai L, Wang X, Xu S, Belinsky SA, et al. Acquired activation of the Akt/cyclooxygenase-2/Mcl-1 pathway renders lung cancer cells resistant to apoptosis. Mol Pharmacol. 2010; 77: 416-423. Ref.: http://bit.ly/2LtjZql
- Li Z, Xu X, Bai L, Chen W, Lin Y. Epidermal growth factor receptor-mediated tissue transglutaminase overexpression couples acquired tumor necrosis factor-related apoptosis-inducing ligand resistance and migration through c-FLIP and MMP-9 proteins in lung cancer cells. J Biol Chem. 2011; 286: 21164-21172. Ref.: http://bit.ly/2XhrX84
- Danial NN, Korsmeyer SJ. Cell death: critical control points. Cell. 2004; 116: 205-219. Ref.: http://bit.ly/323JoMW
- Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000; 100: 57-70. Ref.: http://bit.ly/2NskGCE
- Johnstone RW, Ruefli AA, Lowe SW. Apoptosis: a link between cancer genetics and chemotherapy. Cell. 2002; 108: 153-164. Ref.: http://bit.ly/2ROrOYM
- Fesik SW. Promoting apoptosis as a strategy for cancer drug discovery. Nat Rev Cancer. 2005; 5: 876. Ref.: http://bit.ly/326G83F
- Ashkenazi A. Targeting death and decoy receptors of the tumour-necrosis factor superfamily. Nat Rev Cancer. 2002; 2: 420-430. Ref.: http://bit.ly/2XK6icY
- Falschlehner, C., Emmerich, C.H., Gerlach, B. and Walczak, H., 2007. TRAIL signalling: decisions between life and death. Int J Biochem Cell Biol. 2007; 39: 1462-1475. Ref.: http://bit.ly/2YqbYpC
- Ashkenazi A, Dixit VM. Apoptosis control by death and decoy receptors. Curr Opin Cell Biol. 1999; 11: 255-260. http://bit.ly/2FNcIxE
- Keogh SA, Walczak H, Bouchier-Hayes L, Martin SJ. Failure of Bcl‐2 to block cytochrome c redistribution during TRAIL‐induced apoptosis. FEBS letters. 2000; 471: 93-98. Ref.: http://bit.ly/2RQt8Kt
- Walczak H, Bouchon A, Stahl H, Krammer PH. Tumor necrosis factor-related apoptosis-inducing ligand retains its apoptosis-inducing capacity on Bcl-2-or Bcl-xL-overexpressing chemotherapy-resistant tumor cells. Cancer Res. 2000; 60: 3051-3057. Ref.: http://bit.ly/2FOEr10
- Belka C, Schmid B, Marini P, Durand E, Rudner J, et al. Sensitization of resistant lymphoma cells to irradiation-induced apoptosis by the death ligand TRAIL. Oncogene. 2001; 20: 2190-2196. Ref.: http://bit.ly/2RNyFlf
- Hinz S, Trauzold A, Boenicke L, Sandberg C, Beckmann S, et al. Bcl-XL protects pancreatic adenocarcinoma cells against CD95-and TRAIL-receptor-mediated apoptosis. Oncogene. 2000; 19: 5477-5486. Ref.: http://bit.ly/2XlUvT0
- Fulda S, Meyer E, Debatin KM. Inhibition of TRAIL-induced apoptosis by Bcl-2 overexpression. Oncogene. 2002; 21: 2283. Ref.: http://bit.ly/2RNzfiV
- Zhang XD, Borrow JM, Zhang XY, Nguyen T, Hersey P. Activation of ERK1/2 protects melanoma cells from TRAIL-induced apoptosis by inhibiting Smac/DIABLO release from mitochondria. Oncogene. 2003; 22: Ref.: http://bit.ly/2RKSbyA
- Rudner J, Jendrossek V, Lauber K, Daniel PT, Wesselborg S, et al. Type I and type II reactions in TRAIL-induced apoptosis-results from dose-response studies. Oncogene. 2005; 24: 130-140. Ref.: http://bit.ly/2JjEoLJ
- Secchiero P, Zauli G. Tumor necrosis factor-related apoptosis-inducing ligand and the regulation of hematopoiesis. Curr Opin Hematol. 2008; 15: 42-48. Ref.: http://bit.ly/2NsfpuT
- Janssen EM, Droin NM, Lemmens EE, Pinkoski MJ, Bensinger SJ, et al. CD4+ T-cell help controls CD8+ T-cell memory via TRAIL-mediated activation-induced cell death. Nature. 2005; 434: 88. Ref.: http://bit.ly/2xmWsyP
- Weckmann M, Collison A, Simpson JL, Kopp MV, Wark PA, et al. Critical link between TRAIL and CCL20 for the activation of TH2 cells and the expression of allergic airway disease. Nat Med. 2007; 13: 1308. Ref.: http://bit.ly/2XjAL24
- Cretney E, Shanker A, Yagita H, Smyth MJ, Sayers TJ. TNF-related apoptosis-inducing ligand as a therapeutic agent in autoimmunity and cancer. Immunol Cell Biol. 2006; 84: 87. Ref.: http://bit.ly/2FN9kTE
- Hilliard B, Wilmen A, Seidel C, Liu TS, Göke R, et al. Roles of TNF-related apoptosis-inducing ligand in experimental autoimmune encephalomyelitis. J Immunol. 2001; 166: 1314-1319. Ref.: http://bit.ly/2xmWDdt
- Lamhamedi-Cherradi SE, Zheng S, Tisch RM, Chen YH. Critical roles of tumor necrosis factor–related apoptosis-inducing ligand in type 1 diabetes. Diabetes. 2003; 52: 2274-2278. Ref.: http://bit.ly/2LxUrIG
- Zheng SJ, Wang P, Tsabary G, Chen YH. Critical roles of TRAIL in hepatic cell death and hepatic inflammation. J Clin Invest. 2004; 113: 58-64. Ref.: http://bit.ly/2xoU7nl
- Hoffmann O, Priller J, Prozorovski T, Schulze-Topphoff U, Baeva N, et al. TRAIL limits excessive host immune responses in bacterial meningitis. J Clin Invest. 2007; 117: 2004-2013. Ref.: http://bit.ly/2KRyUuP
- Lancaster JM, Sayer R, Blanchette C, Calingaert B, Whitaker R, et al. High expression of tumor necrosis factor-related apoptosis-inducing ligand is associated with favorable ovarian cancer survival. Clin Cancer Res. 2003; 9: 762-766. Ref.: http://bit.ly/2KRyUuP
- Wright K, Wilson PJ, Kerr J, Do K, Hurst T, et al. Frequent loss of heterozygosity and three critical regions on the short arm of chromosome 8 in ovarian adenocarcinomas. Oncogene. 1998; 17:1185-1188. Ref.: http://bit.ly/2XhEb0H
- Pribill I, Speiser P, Leary J, Leodolter S, Hacker NF, et al. High frequency of allelic imbalance at regions of chromosome arm 8p in ovarian carcinoma. Cancer Genet Cytogenet. 2001; 129: 23-29. Ref.: http://bit.ly/2KRHlq3
- Pan G, Ni J, Wei YF, Yu G, Gentz R, et al. An antagonist decoy receptor and a death domain-containing receptor for TRAIL. Science. 1997; 277: 815-818. Ref.: http://bit.ly/2LxWxZ4
- Lee SH, Shin MS, Kim HS, Lee HK, Park WS, et al. Alterations of the DR5/TRAIL receptor 2 gene in non-small cell lung cancers. Cancer Res. 1999; 59: 5683-5686. Ref.: http://bit.ly/2Lr6ZS2
- Lee SH, Shin MS, Kim HS, Lee HK, Park WS, et al. Somatic mutations of TRAIL-receptor 1 and TRAIL-receptor 2 genes in non-Hodgkin's lymphoma. Oncogene. 2001; 20: 399-403. Ref.: http://bit.ly/2KWycwq
- Fisher MJ, Virmani AK, Wu L, Aplenc R, Harper JC, et al. Nucleotide substitution in the ectodomain of trail receptor DR4 is associated with lung cancer and head and neck cancer. Clin Cancer Res. 2001; 7:1688-1697. Ref.: http://bit.ly/2XHWikq
- Shin MS, Kim HS, Lee SH, Park WS, Kim SY, et al. Mutations of tumor necrosis factor-related apoptosis-inducing ligand receptor 1 (TRAIL-R1) and receptor 2 (TRAIL-R2) genes in metastatic breast cancers. Cancer research 2001; 61: 4942-4946. Ref.: http://bit.ly/2XhNSft
- Bin L, Thorburn J, Thomas LR, Clark PE, Humphreys R, et al. Tumor-derived mutations in the TRAIL receptor DR5 inhibit TRAIL signaling through the DR4 receptor by competing for ligand binding. J Biol Chem. 2007; 282: 28189-28194. http://bit.ly/2Xi62CJ
- MacFarlane M, Kohlhaas SL, Sutcliffe MJ, Dyer MJ, Cohen GM. TRAIL receptor-selective mutants signal to apoptosis via TRAIL-R1 in primary lymphoid malignancies. Cancer Res. 2005; 65: 11265-11270. Ref.: http://bit.ly/305SKWF
- van der Sloot AM, Tur V, Szegezdi E, Mullally MM, Cool RH, Samali A, et al. Designed tumor necrosis factor-related apoptosis-inducing ligand variants initiating apoptosis exclusively via the DR5 receptor. Proc Natl Acad Sci U S A. 2006; 103: 8634-8639. Ref.: http://bit.ly/324Gzvm
- Sanlioglu AD, Karacay B, Koksal IT, Griffith TS, Sanlioglu S. DcR2 (TRAIL-R4) siRNA and adenovirus delivery of TRAIL (Ad5hTRAIL) break down in vitro tumorigenic potential of prostate carcinoma cells. Cancer Gene Ther. 2007; 14: 976-984. Ref.: http://bit.ly/2No8hzI
- Sanlioglu AD, Korcum AF, Pestereli E, Erdogan G, Karaveli S, et al. TRAIL Death Receptor–4 Expression Positively Correlates With the Tumor Grade in Breast Cancer Patients With Invasive Ductal Carcinoma. Int J Radiat Oncol Biol Phys. 2007; 69: 716-723. Ref.: http://bit.ly/2RU8kC8
- Riccioni R, Pasquini L, Mariani G, Saulle E, Rossini A, et al. TRAIL decoy receptors mediate resistance of acute myeloid leukemia cells to TRAIL. Haematologica. 2005; 90: 612-624. Ref.: http://bit.ly/2Xi7yEV
- Mérino D, Lalaoui N, Morizot A, Schneider P, Solary E, et al. Differential inhibition of TRAIL-mediated DR5-DISC formation by decoy receptors 1 and 2. Mol Cell Biol. 2006; 26: 7046-7055. Ref.: http://bit.ly/30eCRh1
- Emery JG, McDonnell P, Burke MB, Deen KC, Lyn S, et al. Osteoprotegerin is a receptor for the cytotoxic ligand TRAIL. J Biol Chem. 1998; 273: 14363-14367. Ref.: http://bit.ly/2YsMXdl
- Devin A, Cook A, Lin Y, Rodriguez Y, Kelliher M, et al. The distinct roles of TRAF2 and RIP in IKK activation by TNF-R1: TRAF2 recruits IKK to TNF-R1 while RIP mediates IKK activation. Immunity. 2000; 12: 419-429. Ref.: http://bit.ly/2XCTJ3e
- Yeh WC, Shahinian A, Speiser D, Kraunus J, Billia F, et al. Early lethality, functional NF-κB activation, and increased sensitivity to TNF-induced cell death in TRAF2-deficient mice. Immunity. 1997; 7: 715-725. Ref.: http://bit.ly/2XoEdbN
- Hsu H, Huang J, Shu HB, Baichwal V, Goeddel DV. TNF-dependent recruitment of the protein kinase RIP to the TNF receptor-1 signaling complex. Immunity. 1996; 4:387-396. Ref.: http://bit.ly/2Xhytfg
- Nagata S. Fas ligand-induced apoptosis. Annu Rev Genet.1999; 33: 29-55. Ref.: http://bit.ly/2YsNdsP
- Peter ME, Krammer PH. The CD95 (APO-1/Fas) DISC and beyond. Cell Death Differ. 2003; 10: 26-35. Ref.: http://bit.ly/2YnfX6p
- Song JH, Tse MC, Bellail A, Phuphanich S, Khuri F, et al. Lipid rafts and nonrafts mediate tumor necrosis factor–related apoptosis-inducing ligand–induced apoptotic and nonapoptotic signals in non–small cell Lung Carcinoma Cells. Cancer Res. 2007; 67: 6946-6955. Ref.: http://bit.ly/2J4J35m
- Boldin MP, Goncharov TM, Goltsev YV, Wallach D. Involvement of MACH, a novel MORT-IIFADD-interacting protease. Fas/Apo-l. 1996; 803-815.
- Chinnaiyan AM1, Tepper CG, Seldin MF, O'Rourke K, Kischkel FC, et al. FADD/MORT1 is a common mediator of CD95 (Fas/APO-1) and tumor necrosis factor receptor-induced apoptosis. J Biol Chem. 1996; 271: 4961-4965. Ref.: http://bit.ly/2NuxCbk
- MacFarlane M. TRAIL-induced signalling and apoptosis. Toxicol Lett. 2003; 139: 89-97. Ref.: http://bit.ly/2Jnrmg3
- Degli-Esposti MA, Dougall WC, Smolak PJ, Waugh JY, Smith CA, et al. The novel receptor TRAIL-R4 induces NF-κB and protects against TRAIL-mediated apoptosis, yet retains an incomplete death domain. Immunity. 1997; 7: 813-820. Ref.: http://bit.ly/2LFh2mL
- Li H, Lin X. Positive and negative signaling components involved in TNFα-induced NF-κB activation. Cytokine. 2008; 41: 1-8. Ref.: http://bit.ly/308Su9l
- Trauzold A, Siegmund D, Schniewind B, Sipos B, Egberts J, et al. TRAIL promotes metastasis of human pancreatic ductal adenocarcinoma. Oncogene. 2006; 25: 7434. Ref.: http://bit.ly/2KSnGq4
- Ricci MS, Kim SH, Ogi K, Plastaras JP, Ling J, et al. Reduction of TRAIL-induced Mcl-1 and cIAP2 by c-Myc or sorafenib sensitizes resistant human cancer cells to TRAIL-induced death. Cancer Cell. 2007; 12: 66-80. Ref.: http://bit.ly/2J5frEL
- Secchiero P, Gonelli A, Carnevale E, Milani D, Pandolfi A, et al. TRAIL promotes the survival and proliferation of primary human vascular endothelial cells by activating the Akt and ERK pathways. Circulation. 2003; 107: 2250-2256. Ref.: http://bit.ly/2LyCkSD
Figures:

Figure 1
Similar Articles
-
Insilico investigation of TNFSF10 signaling cascade in ovarian serous cystadenocarcinomaAsima Tayyeb*,Zafar Abbas Shah. Insilico investigation of TNFSF10 signaling cascade in ovarian serous cystadenocarcinoma. . 2019 doi: 10.29328/journal.acst.1001005; 3: 025-034
Recently Viewed
-
From Wallets to Waistlines: Exploring the Link between Family Income (Wage Distribution) and Obesity Indices in Pakistani PopulationsAtif Akbar,Waqas Ghulam Hussain*. From Wallets to Waistlines: Exploring the Link between Family Income (Wage Distribution) and Obesity Indices in Pakistani Populations. New Insights Obes Gene Beyond. 2026: doi: 10.29328/journal.niogb.1001024; 10: 001-014
-
Screening for Depressive Symptoms in Clinical and Nonclinical Youth: The Psychometric Properties of the Dutch Children’s Depression Inventory-2 (CDI-2)Denise HM Bodden*,Yvonne Stikkelbroek,Daan Creemers,Sanne PA Rasing,Elien De Caluwe,Caroline Braet. Screening for Depressive Symptoms in Clinical and Nonclinical Youth: The Psychometric Properties of the Dutch Children’s Depression Inventory-2 (CDI-2). Insights Depress Anxiety. 2025: doi: 10.29328/journal.ida.1001047; 9: 028-039
-
Knowledge, Attitude, and Practice of Healthcare Workers in Ekiti State, Nigeria on Prevention of Cervical CancerAde-Ojo Idowu Pius*, Okunola Temitope Omoladun, Olaogun Dominic Oluwole. Knowledge, Attitude, and Practice of Healthcare Workers in Ekiti State, Nigeria on Prevention of Cervical Cancer. Arch Cancer Sci Ther. 2024: doi: 10.29328/journal.acst.1001038; 8: 001-006
-
Oral Cancer Management is not just Treatment! But also, how early Pre-cancerous Lesions are Diagnosed & Treated!!Suresh Kishanrao*. Oral Cancer Management is not just Treatment! But also, how early Pre-cancerous Lesions are Diagnosed & Treated!!. Arch Cancer Sci Ther. 2024: doi: 10.29328/journal.acst.1001039; 8: 007-012
-
Breast Cancer in FemaleLorena Menditto*. Breast Cancer in Female. Arch Cancer Sci Ther. 2024: doi: 10.29328/journal.acst.1001040; 8: 013-018
Most Viewed
-
Physical Performance in the Overweight/Obesity Children Evaluation and RehabilitationCristina Popescu, Mircea-Sebastian Șerbănescu, Gigi Calin*, Magdalena Rodica Trăistaru. Physical Performance in the Overweight/Obesity Children Evaluation and Rehabilitation. Ann Clin Endocrinol Metabol. 2024 doi: 10.29328/journal.acem.1001030; 8: 004-012
-
Hypercalcaemic Crisis Associated with Hyperthyroidism: A Rare and Challenging PresentationKarthik Baburaj*, Priya Thottiyil Nair, Abeed Hussain, Vimal MV. Hypercalcaemic Crisis Associated with Hyperthyroidism: A Rare and Challenging Presentation. Ann Clin Endocrinol Metabol. 2024 doi: 10.29328/journal.acem.1001029; 8: 001-003
-
Effects of dietary supplementation on progression to type 2 diabetes in subjects with prediabetes: a single center randomized double-blind placebo-controlled trialSathit Niramitmahapanya*,Preeyapat Chattieng,Tiersidh Nasomphan,Korbtham Sathirakul. Effects of dietary supplementation on progression to type 2 diabetes in subjects with prediabetes: a single center randomized double-blind placebo-controlled trial. Ann Clin Endocrinol Metabol. 2023 doi: 10.29328/journal.acem.1001026; 7: 00-007
-
Exceptional cancer responders: A zone-to-goDaniel Gandia,Cecilia Suárez*. Exceptional cancer responders: A zone-to-go. Arch Cancer Sci Ther. 2023 doi: 10.29328/journal.acst.1001033; 7: 001-002
-
Ectopic adrenal tissue at the spermatic cordAbdallah Chaachou,Nizar Cherni,Wael Ferjaoui*,Mohamed Dridi,Samir Ghozzi. Ectopic adrenal tissue at the spermatic cord. J Clin Med Exp Images. 2022 doi: 10.29328/journal.jcmei.1001024; 6: 001-002

If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."