More Information
Submitted: March 22, 2026 | Approved: March 31, 2026 | Published: April 01, 2026
How to cite this article: Elafari MA, Ayoub M, Bibat MA, Slaoui A, Karmouni T, Koutani A, et al. Late Extensive Regional Recurrence of Paratesticular Dedifferentiated Liposarcoma 10 Years after Orchiectomy: A Case Report. Arch Cancer Sci Ther. 2026; 10(1): 006-010. Available from:
https://dx.doi.org/10.29328/journal.acst.1001049
DOI: 10.29328/journal.acst.1001049
Copyright license: © 2026 Elafari MA, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Paratesticular liposarcoma; Dedifferentiated liposarcoma; Late recurrence; MDM2; Neoadjuvant chemotherapy
Late Extensive Regional Recurrence of Paratesticular Dedifferentiated Liposarcoma 10 Years after Orchiectomy: A Case Report
Mohammed Amine Elafari* , Mamad Ayoub, Mohammed Amine Bibat, Amine Slaoui, Tariq Karmouni, Abdelatif Koutani and Khalid Elkhader
, Mamad Ayoub, Mohammed Amine Bibat, Amine Slaoui, Tariq Karmouni, Abdelatif Koutani and Khalid Elkhader
Urology B Department, IBN SINA Hospital, University Hospital Center IBN SINA, University Mohammed V, Rabat, Morocco
*Address for Correspondence: Mohammed Amine Elafari, Urology B Department, IBN SINA Hospital, University Hospital Center IBN SINA, University Mohammed V, Rabat, Morocco, Email: [email protected]
Background: Paratesticular liposarcoma is a rare entity, with dedifferentiated liposarcoma (DDLPS) representing the most aggressive subtype. Late recurrences with unusual patterns of spread have been documented but remain poorly characterized.
Case presentation: We report a case of a 68-year-old male with a 10-year recurrence of paratesticular DDLPS following right orchiectomy. The patient initially underwent resection of a 5×4×4.5 cm paratesticular mass in 2015, with histopathology confirming DDLPS and negative distal spermatic cord margins. After loss to follow-up, he presented in 2026 with extensive regional recurrence involving bilateral pubic soft tissues, abdominal wall musculature, and right external iliac lymphadenopathy. Biopsy confirmed recurrent DDLPS with Mouse double minute 2 homolog (MDM2) positivity. Given the multi-site infiltrative nature and muscle involvement, the multidisciplinary team recommended neoadjuvant chemotherapy with ifosfamide and doxorubicin before consideration of surgical resection.
Conclusion: This case illustrates the propensity for late regional recurrence in paratesticular DDLPS and highlights the unusual pattern of spread along anatomic pathways, including the abdominal wall and inguinal region. The case underscores the importance of long-term surveillance, aggressive initial surgical margins, and multidisciplinary management in this rare malignancy.
Paratesticular liposarcoma is an exceedingly rare entity, accounting for less than 1% of all genitourinary cancers and approximately 7% of all paratesticular sarcomas. Globally, the incidence is estimated at 0.2 per million persons per year, with a peak occurrence in the fifth to sixth decades of life. Dedifferentiated liposarcoma (DDLPS) represents the most aggressive subtype, characterized by the transformation of well-differentiated areas into high-grade, non-lipogenic sarcoma. Clinically, it most frequently presents as a painless, firm, and slow-growing inguinal or scrotal mass, which often leads to misdiagnosis as more common benign conditions such as inguinal hernias, hydroceles, or epididymitis [1-3]. DDLPS is identified by amplification of the Mouse double minute 2 homolog (MDM2) and CDK4 genes located at chromosome 12q13-15 and acts as a diagnostic hallmark for this entity [3-5].
The retroperitoneal area and deep tissues of the extremities are most commonly affected by DDLPS, and paratesticular involvement is extremely rare [2,4]. Paratesticular liposarcoma presents as painless, slow-growing inguinal or scrotal masses and is most commonly misinterpreted as benign conditions such as hernias or hydroceles [6-8]. The recommended therapy for this entity is radical orchiectomy along with high inguinal ligation of the spermatic cord to ensure negative margins, and positive margins are identified as the strongest predictor for local recurrence [6,7,9].
The pattern of recurrence for paratesticular DDLPS is unique, where local recurrence is more common compared to distant metastasis [6,10]. Interestingly, local recurrence occurs along the gonadal vessels and involves the inguinal canal, retroperitoneal space, and abdominal wall [6]. Furthermore, late recurrences occurring years to decades after initial resection are also reported, emphasizing the need for long-term follow-up [10].
We report this rare case of paratesticular DDLPS, where the patient presented with extensive regional recurrence in the bilateral pubic soft tissues, abdominal wall musculature, and regional lymphadenopathy after a 10-year interval following initial resection, thus highlighting the biological behavior of this rare malignancy.
A 68-year-old male with an 8-year history of diabetes mellitus presented with multiple palpable masses in the pubic and inguinal regions in early 2026.
His oncologic history was significant for a right orchiectomy performed in 2015 at age 57 for a voluminous paratesticular mass. Gross examination at that time revealed a heterogeneous soft-tissue tumor measuring 5×4×4.5 cm, located in contact with the tunica albuginea. Histopathological analysis demonstrated a fasciculated tumoral proliferation with high-grade nuclear atypia and numerous mitoses, consistent with dedifferentiated liposarcoma. Although the distal resection margin of the spermatic cord was reported as negative, the patient remained lost to medical surveillance for 10 years. This prolonged absence was primarily due to the patient’s perceived clinical recovery and the absence of symptoms, combined with socio-economic constraints that hindered his return to the oncology center for scheduled imaging.
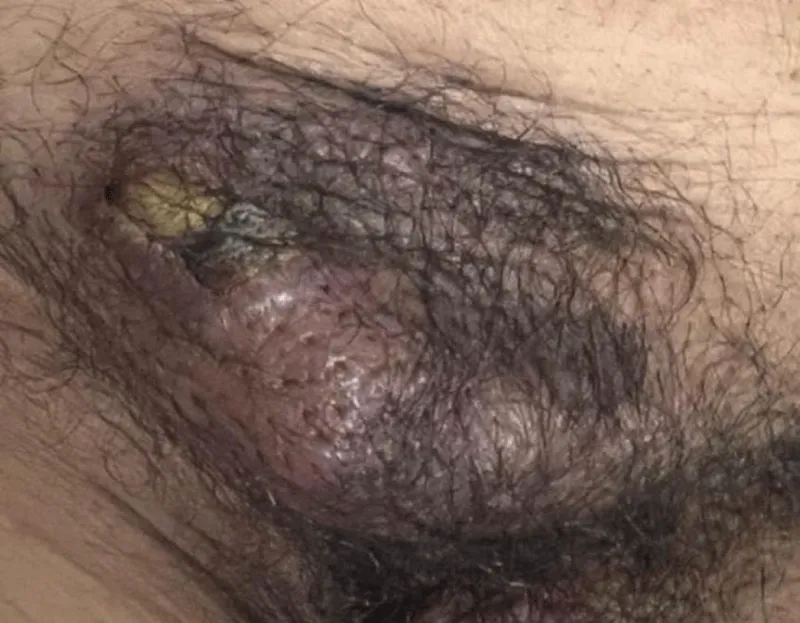
Ten years after the initial surgery, the patient returned with multiple palpable, infiltrating masses in the pubic and inguinal regions (Figure 1).
Figure 1: Clinical presentation of the regional recurrence.
The image shows multiple, firm, infiltrating subcutaneous masses in the pubic and bilateral inguinal regions appearing 10 years after the initial right orchiectomy.
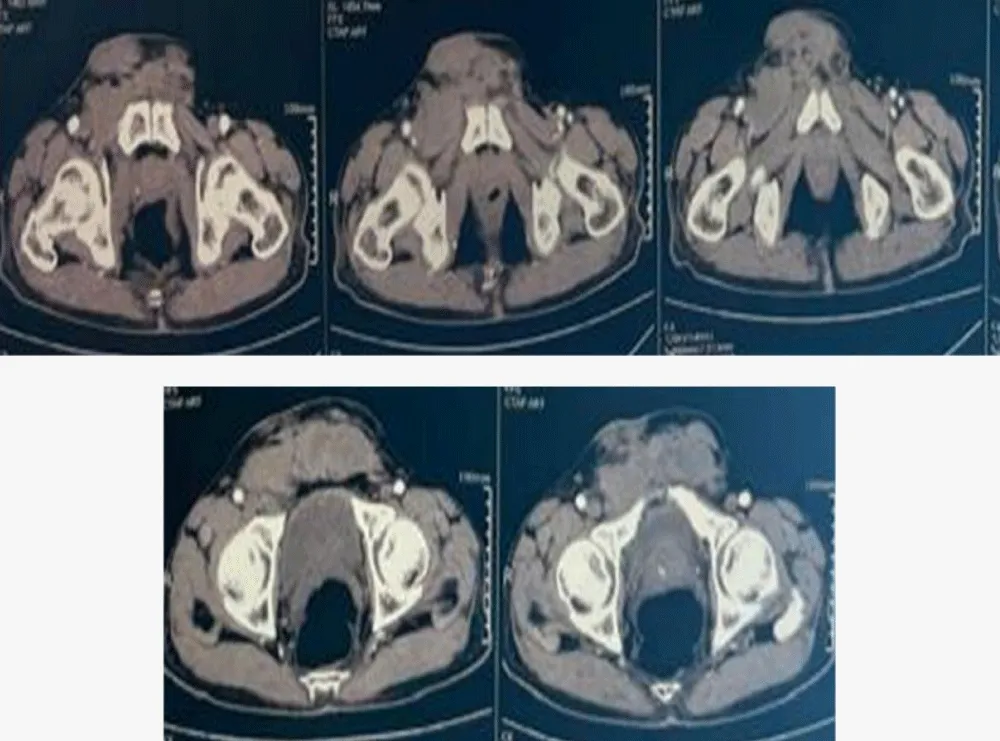
Computed tomography (CT) of the chest, abdomen, and pelvis performed in December 2025 (portal venous phase, 2.5 mm slice thickness) demonstrated extensive regional recurrence characterized by multiple infiltrating soft-tissue masses in the pubic region, including a 25×42 mm mass involving the right transverse abdominis muscle with infiltration of the ipsilateral pectineus muscle, a 34×72 mm mass infiltrating both rectus abdominis muscles, subcutaneous pubic masses measuring 49×26 mm on the right and 30×20 mm on the left, and right external iliac lymphadenopathy measuring 26×39 mm, without evidence of bone involvement, intraperitoneal spread, or pulmonary metastases (Figure 2).
Figure 2: Contrast-enhanced axial CT scan of the pelvis (portal phase, 2.5 mm slice thickness).
The imaging demonstrates extensive regional recurrence characterized by multiple infiltrating soft-tissue masses, including a 34×72 mm mass involving both rectus abdominis muscles and the right transverse abdominis muscle. There is notable involvement of the right external iliac lymph nodes (26×39 mm) and bilateral subcutaneous pubic masses, following the anatomical pathways of the initial surgical site and spermatic cord.
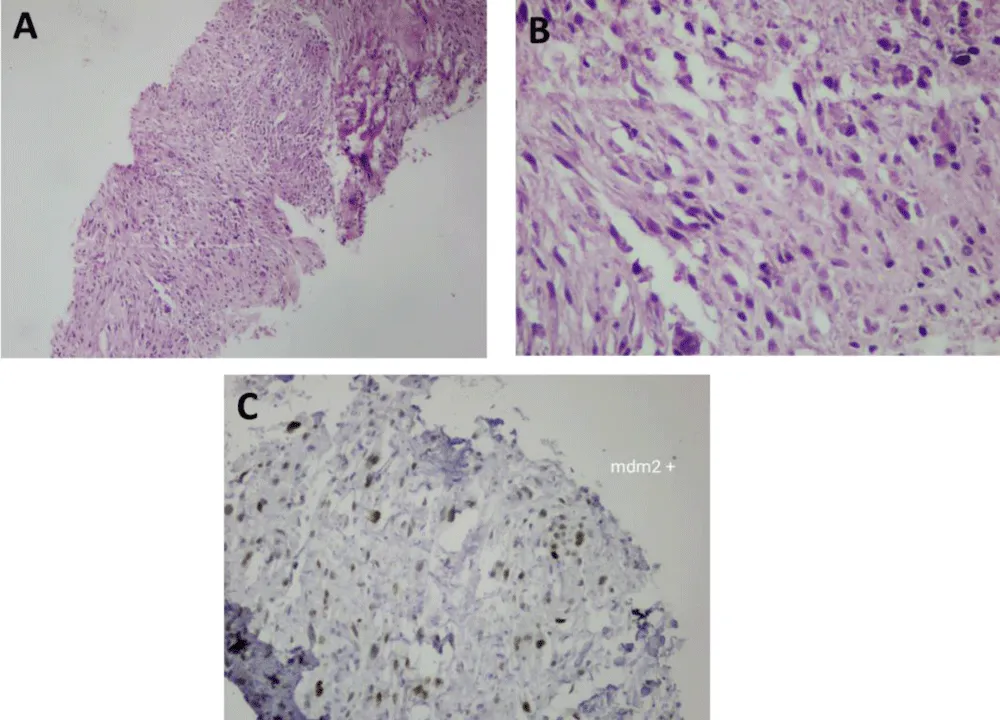
Biopsy of the pubic masses in January 2026 demonstrated a spindle-cell tumor with a mitotic count of 1 per 10 high-power fields (HPF) and approximately 50% focal necrosis. This high mitotic activity and the presence of necrosis supported the diagnosis of recurrent DDLPS over a purely well-differentiated lesion. Immunohistochemistry revealed strong and diffuse nuclear positivity for MDM2, confirming the diagnosis of recurrent liposarcoma. The morphologic and immunophenotypic profile was consistent with either sclerosing well-differentiated liposarcoma or low-grade dedifferentiated liposarcoma (Figure 3).
Figure 3: Histopathological and Immunohistochemical Analysis. (A) H&E, x100: Fasciculated mesenchymal proliferation infiltrating soft tissues. (B) H&E, x400: High-grade spindle cells with hyperchromatic nuclei and brisk mitotic activity (1/10 HPF). (C) IHC, x400: Strong and diffuse nuclear positivity for MDM2, confirming the diagnosis.
The case was presented at a multidisciplinary tumor board. Given the multi-site infiltrative nature of the recurrence, involvement of bilateral abdominal wall muscles, and regional lymphadenopathy, the tumor was deemed technically challenging for upfront surgical resection. The multidisciplinary team recommended neoadjuvant chemotherapy. The planned regimen consists of four cycles of Doxorubicin (75 mg/m² as a continuous 72-hour infusion) and Ifosfamide (9 g/m² divided over 3 days), administered every 21 days along with Mesna for uroprotection to prevent hemorrhagic cystitis, following the standard of care for high-grade soft tissue sarcomas to achieve tumor shrinkage and facilitate subsequent radical surgical resection.
Following surgical resection, adjuvant radiotherapy (50-60 Gy) will be considered to enhance local control, particularly if R1 margins are encountered. Furthermore, should the disease progress, targeted therapies such as CDK4/6 inhibitors (e.g., palbociclib) may be utilized, given the patient’s MDM2/CDK4 amplification status.
This case highlights some of the significant characteristics of paratesticular DDLPS, which include the risk of late regional recurrence, the pattern of recurrence, and the difficulties in the management of recurrent cases of locally advanced DDLPS.
Paratesticular liposarcomas have a unique pattern of recurrence compared to those in other sites. Khandekar, et al. studied 25 patients with paratesticular liposarcoma and noted the 3-year and 5-year local recurrence-free survival rates to be 76% and 67%, respectively [6]. What is noteworthy is that the study noted the adverse impact of positive surgical margins and recurrent cases on local recurrence [6,9]. Although in the present case, the margins of the distal spermatic cord showed no tumor, the recurrence was widespread in the 10-year follow-up.
The pattern of recurrence in this patient, which involves bilateral pubic soft tissues, abdominal wall muscles, and regional lymph nodes, is in keeping with previously described patterns of recurrence along the pathways of gonadal vessels and other anatomic pathways [6]. In fact, Khandekar, et al. documented that recurrences in paratesticular liposarcoma follow along the pathways of the gonadal vessels and should be taken into consideration in surveillance imaging [6]. The involvement of abdominal wall muscles and inguinal lymph nodes in our patient is in keeping with tumor spread along the spermatic cord pathway and regional lymphatic spread.
Recent studies (2022-2025) confirm that while most recurrences occur within the first 2-5 years, paratesticular DDLPS maintains a lifelong risk of regional spread. Our case is comparable to the series by Montgomery and Fisher, which documented rare recurrences up to 21 years post-orchiectomy, but the extensive multi-site involvement (bilateral abdominal wall and musculature) seen here after a 10-year silent interval is particularly unusual [10].
Dedifferentiated liposarcoma has a poorer outcome than the well-differentiated types. Studies have shown that the population outcome for DDLPS has a median survival of 54-70 months, and 5-year survival is approximately 48% [1,11]. Poor outcomes are independently predicted by advanced age, tumor size, stage, and retroperitoneal primary site [1,12,13]. Although paratesticular DDLPS is less common than retroperitoneal DDLPS, it has similar clinical behavior with a tendency to local recurrence and, less frequently, distant metastasis [6,10].
The presence of MDM2 amplification, which was confirmed by immunohistochemistry in the current case, is a characteristic feature of DDLPS and helps to distinguish it from other aggressive sarcomas [3-5]. MDM2 amplification is due to the presence of supernumerary ring and giant marker chromosomes with amplified sequences of chromosome 12q13-15 [4,14].
The role of neoadjuvant chemotherapy in DDLPS has been questioned, with limited data available for this particular site. For high-risk soft tissue sarcomas, as in DDLPS, anthracyclines with or without ifosfamide are standard first-line systemic treatments [4,15,16].
The ISG-ST S1001 trial has shown that neoadjuvant chemotherapy with full-dose anthracyclines and ifosfamide improves disease-free and overall survival in high-risk soft tissue sarcomas of the extremities and trunk [17].
In the context of advanced or locally recurrent DDLPS, doxorubicin and ifosfamide are active with response rates ranging from 6 to 22% [15,18]. Goy, et al. have shown that ifosfamide-doxorubicin-based chemotherapy independently predicted improved distant metastasis-free survival, disease-specific survival, and overall survival in high-grade soft tissue sarcomas, including DDLPS [18]. The response rates to these agents, however, remain low, and the role of neoadjuvant chemotherapy in facilitating resection must be set against the possible delay in definitive treatment [15,17].
The current NCCN guidelines suggest anthracycline-based regimens, including doxorubicin/ifosfamide/mesna, as the preferred neoadjuvant therapy for high-risk STS [16]. For DDLPS, systemic therapies include anthracycline-based regimens, gemcitabine-based regimens, eribulin, trabectedin, and CDK4/6 inhibitors, including palbociclib and abemaciclib [16].
In this case, the decision to administer neoadjuvant chemotherapy was based on the extensive disease, including the infiltration of the bilateral abdominal wall muscles, and the technical difficulties in achieving a complete resection with negative margins. The idea behind administering neoadjuvant chemotherapy is to induce tumor shrinkage, thus facilitating a potentially curative resection.
Surgical resection with negative margins is the cornerstone in the treatment of localized paratesticular liposarcoma [6,7,9]. High inguinal orchiectomy with en bloc resection of the spermatic cord is the recommended surgical technique, as it has been associated with a higher recurrence-free survival compared to simple tumorectomy [7]. The most significant prognostic factor for recurrence is the presence of positive surgical margins, and wide re-resection should be attempted when feasible [7,9].
In the present case, the extensive infiltration of the bilateral rectus abdominis and transverse muscles presents a formidable surgical challenge. Achieving a microscopically margin-negative resection (R0 resection) after neoadjuvant treatment will likely require a complex abdominal wall reconstruction using prosthetic mesh to maintain structural integrity. As noted in the literature, achieving such margins in cases with significant regional involvement may necessitate a radical procedure involving hemiscrotectomy, wide abdominal wall resection, and lymphadenectomy [9].
Furthermore, the tumor’s proximity to major pelvic vessels and structures may require a radical ‘salvage’ procedure, underscoring the aggressive biological behavior of DDLPS. This subtype tends to spread along anatomical planes and the spermatic cord pathway, making secondary surgical control significantly more difficult than the primary intervention. While aggressive surgery is the cornerstone of management, the role of adjuvant radiotherapy in paratesticular liposarcoma remains debatable; some studies indicate little to no significant benefit, even in the presence of positive margins [6,7].
Considering the risk of late recurrence in paratesticular liposarcoma, follow-up is critical. The NCCN guidelines suggest that the primary site should be evaluated with a physical examination and imaging studies at 6-12 month intervals for 2 years, followed by yearly evaluations, and the frequency of follow-up should be determined based on the risk of recurrence at the primary site [16]. The follow-up should include images that are useful in detecting recurrence in the spermatic cord, inguinal canal, retroperitoneum, and abdominal wall [6]. Chest images are not necessary in the follow-up of patients with well-differentiated liposarcoma, but may be necessary in the follow-up of patients with dedifferentiated liposarcoma [16].
This case illustrates the difficulties encountered in the management of paratesticular dedifferentiated liposarcoma, a rare tumor with a tendency for late regional recurrence and unpredictable metastasis. The patient had a large regional recurrence 10 years following the initial tumor resection with negative margins, affecting the bilateral pubic soft tissues, abdominal wall muscles, and lymph node metastasis. This case emphasizes the need for aggressive initial resection of the tumor with negative margins. The management of the tumor with neoadjuvant chemotherapy with ifosfamide and doxorubicin was undertaken to make subsequent surgical resection easier for the patient with advanced recurrent disease. The long-term outcome of the disease will be influenced by the response to systemic treatment and the ability to achieve a complete resection with negative margins.
Declaration
Ethical approval: Ethics approval has been obtained to proceed with the current study.
Ethical approval for this study (Ethical Committee N009-24) was provided by the Ethical Committee of Ibn University Hospitals, Rabat, Morocco, on 22 January 2024.
Consent: Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of the journal.
Guarantor of submission: The corresponding author is the guarantor of the submission.
Availability of data and materials: Supporting material is available if further analysis is needed.
Provenance and peer review: Not commissioned, externally peer-reviewed.
- Khandekar MJ, Raut CP, Hornick JL, Wang Q, Alexander BM, Baldini EH. Paratesticular liposarcoma: unusual patterns of recurrence and importance of margins. Ann Surg Oncol. 2013;20(7):2148-2155. Available from: https://doi.org/10.1245/s10434-013-2963-0
- Kamitani R, Matsumoto K, Takeda T, Mizuno R, Oya M. Optimal treatment strategy for paratesticular liposarcoma: retrospective analysis of 265 reported cases. Int J Clin Oncol. 2020;25(12):2099-2106. Available from: https://doi.org/10.1007/s10147-020-01753-3
- Obeidat A. Predictors of survival in dedifferentiated liposarcoma: a population-based analysis of the SEER database. Medicine (Baltimore). 2026;105(9):e47738. Available from: https://doi.org/10.1097/md.0000000000047738
- Lee ATJ, Thway K, Huang PH, Jones RL. Clinical and molecular spectrum of liposarcoma. J Clin Oncol. 2018;36(2):151-159. Available from: https://doi.org/10.1200/jco.2017.74.9598
- Thway K. Well-differentiated liposarcoma and dedifferentiated liposarcoma: an updated review. Semin Diagn Pathol. 2019;36(2):112-121. Available from: https://doi.org/10.1053/j.semdp.2019.02.006
- Nishio J, Nakayama S, Nabeshima K, Yamamoto T. Biology and management of dedifferentiated liposarcoma: state of the art and perspectives. J Clin Med. 2021;10(15):3230. Available from: https://doi.org/10.3390/jcm10153230
- Haddox CL, Hornick JL, Roland CL, Baldini EH, Keedy VL, Riedel RF. Diagnosis and management of dedifferentiated liposarcoma: a multidisciplinary position statement. Cancer Treat Rev. 2024;131:102846. Available from: https://doi.org/10.1016/j.ctrv.2024.102846
- Li J, Wang J, Han H, Tian L, Yin H. Case report: two cases of primary paratesticular liposarcoma. Front Oncol. 2022;12:1040458. Available from: https://doi.org/10.3389/fonc.2022.1040458
- Reback T, Pang KH, Haider A, Freeman A, Shankar A, Alnajjar HM, et al. Paratesticular sarcoma: analysis of oncological outcomes and prognostic factors. Clin Genitourin Cancer. 2025;23(3):102331. Available from: https://doi.org/10.1016/j.clgc.2025.102331
- Montgomery E, Fisher C. Paratesticular liposarcoma: a clinicopathologic study. Am J Surg Pathol. 2003;27(1):40-47. Available from: https://doi.org/10.1097/00000478-200301000-00005
- Zhao J, Du W, Tao X, Li A, Li Y, Zhang S. Survival and prognostic factors among different types of liposarcomas based on the SEER database. Sci Rep. 2025;15(1):1790. Available from: https://doi.org/10.1038/s41598-025-85937-9
- Casier J, Timmermans I, Laenen A, Hompes D, Douchy T, Sciot R, et al. Clinical course and prognostic factors of patients with dedifferentiated liposarcoma: a retrospective analysis. BMC Cancer. 2025;25(1):517. Available from: https://doi.org/10.1186/s12885-025-13813-w
- Mori T, Yamada Y, Kinoshita I, Kohashi K, Yamamoto H, Ito Y, et al. Clinicopathological and histopathological review of dedifferentiated liposarcoma: a comprehensive study of 123 primary tumours. Histopathology. 2022;80(3):538-557. Available from: https://doi.org/10.1111/his.14588
- Thway K, Jones RL, Noujaim J, Zaidi S, Miah AB, Fisher C. Dedifferentiated liposarcoma: updates on morphology, genetics, and therapeutic strategies. Adv Anat Pathol. 2016;23(1):30-40. Available from: https://doi.org/10.1097/pap.0000000000000101
- Stacchiotti S, Van der Graaf WTA, Sanfilippo RG, Marreaud SI, Van Houdt WJ, Judson IR, et al. First-line chemotherapy in advanced intra-abdominal well-differentiated/dedifferentiated liposarcoma: an EORTC Soft Tissue and Bone Sarcoma Group retrospective analysis. Cancer. 2022;128(15):2932-2938. Available from: https://doi.org/10.1002/cncr.34264
- National Comprehensive Cancer Network (NCCN). Soft tissue sarcoma (Version 1.2026). 2026. Available from: https://www.nccn.org
- Gronchi A, Ferrari S, Quagliuolo V, Broto JM, Pousa AL, Grignani G, et al. Histotype-tailored neoadjuvant chemotherapy versus standard chemotherapy in patients with high-risk soft-tissue sarcomas (ISG-STS 1001): an international, open-label, randomised, controlled, phase 3, multicentre trial. Lancet Oncol. 2017;18(6):812-822. Available from: https://doi.org/10.1016/s1470-2045(17)30334-0
- Goy BW, Syed S, Padmanabhan A, Burchette RJ, Helmstedter CS. The role of ifosfamide-doxorubicin chemotherapy in histology-specific, high-grade, locally advanced soft tissue sarcoma: a 14-year experience. Radiother Oncol. 2021;165:174-178. Available from: https://doi.org/10.1016/j.radonc.2021.10.019