More Information
Submitted: March 27, 2026 | Approved: April 07, 2026 | Published: April 08, 2026
How to cite this article: Elafari MA, Ayoub M, Bibat MA, Slaoui A, Karmouni T, Koutani A, et al. Urinary Biomarkers: Is the Era of Exclusive Follow-Up Cystoscopy Coming to an End? A Mini-Review of Emerging Molecular Diagnostics and Risk-Stratified Surveillance. Arch Cancer Sci Ther. 2026; 10(1): 011-015. Available from:
https://dx.doi.org/10.29328/journal.acst.1001050
DOI: 10.29328/journal.acst.1001050
Copyright license: © 2026 Elafari MA, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Paratesticular liposarcoma; Dedifferentiated liposarcoma; Late recurrence; MDM2; Neoadjuvant chemotherapy
Urinary Biomarkers: Is the Era of Exclusive Follow-Up Cystoscopy Coming to an End? A Mini-Review of Emerging Molecular Diagnostics and Risk-Stratified Surveillance
Mohammed Amine Elafari* , Mamad Ayoub, Mohammed Amine Bibat, Amine Slaoui, Tariq Karmouni, Abdelatif Koutani and Khalid Elkhader
, Mamad Ayoub, Mohammed Amine Bibat, Amine Slaoui, Tariq Karmouni, Abdelatif Koutani and Khalid Elkhader
Urology B Department, IBN SINA Hospital, University Hospital Center IBN SINA, University Mohammed V, Rabat, Morocco
*Address for Correspondence: Mohammed Amine Elafari, Urology B Department, IBN SINA Hospital, University Hospital Center IBN SINA, University Mohammed V, Rabat, Morocco, Email: [email protected]
Bladder cancer is among the most costly malignancies to treat, primarily due to the high rate of recurrence for Non-Muscle-Invasive Bladder Cancer (NMIBC) and the subsequent necessity for lifelong cystoscopic surveillance. Although cystoscopy is widely regarded as the gold standard, it is an invasive procedure that can be costly and is associated with patient discomfort and anxiety. In recent years, a novel generation of urinary biomarkers (comprising genomic, epigenomic, and proteomic assays) has emerged, exhibiting diagnostic capabilities that increasingly challenge the prevailing role of cystoscopy in surveillance protocols. This mini-review examines the landscape of FDA-approved and emerging urinary biomarkers, compares their diagnostic accuracy, evaluates the first randomized controlled trials supporting biomarker-guided surveillance, and proposes a risk-stratified algorithm for the future integration of urinary biomarkers into clinical practice.
Bladder cancer is the 10th most prevalent malignancy worldwide, with approximately 75% of cases manifesting as NMIBC [1]. The hallmark of NMIBC is its proclivity for recurrence, with up to 60% of patients experiencing recurrence within the first year of diagnosis [2]. Current guidelines from the NCCN, AUA/SUO, and EAU mandate meticulous cystoscopic surveillance at intervals determined by risk stratification, ranging from every 3 months for high-risk disease to annually for low-risk disease [3,4]. This surveillance paradigm imposes a substantial burden on patients and healthcare systems alike.
Cystoscopy, while highly sensitive for papillary tumors, has recognized limitations. It is operator-dependent, may miss flat lesions such as Carcinoma in situ (CIS), and carries procedural morbidity, including dysuria, hematuria, urinary tract infection, and significant patient anxiety [1,5]. These limitations have fueled a decades-long search for noninvasive alternatives.
The current standard: cystoscopy and urine cytology
Cystoscopy is widely regarded as the gold standard for the surveillance of NMIBC. According to the NCCN 2026 guidelines, “office cystoscopy at recommended intervals is a critical diagnostic and surveillance tool for NMIBC,” and that “omission or delay of cystoscopy is acceptable only if a predictive biomarker has been tested and found to be non-inferior in a prospective randomized clinical trial” [3].
Urine cytology has long served as the primary noninvasive adjunct. The test demonstrates an 89–99% specificity for high-grade urothelial carcinoma, yet it exhibits suboptimal sensitivity, particularly for low-grade tumors (13–42%) [2,6,7]. The National Comprehensive Cancer Network (NCCN) guidelines currently advocate for urine cytology in conjunction with cystoscopy for intermediate- and high-risk Non-Muscle-Invasive Bladder Cancer (NMIBC), while acknowledging a lack of a routine role for cytology or biomarkers in low-risk disease [3].
FDA-approved urinary biomarkers
Several urinary biomarker tests have received FDA approval for the detection or monitoring of bladder cancer:
NMP22, or Nuclear Matrix Protein 22, is a protein that plays a role in the formation of the nuclear membrane. The assay is available in the form of a quantitative enzyme-linked immunosorbent assay (ELISA) and a qualitative point-of-care test (BladderChek). The sensitivity of this test ranges from 10–40%, depending on the tumor grade, with a specificity of approximately 95% [6,8].
BTA (Bladder Tumor Antigen) is available in the form of both BTA stat (qualitative) and BTA TRAK (quantitative). The sensitivity for low-grade recurrence is approximately 60%, with a specificity of 51% [6,8].
UroVysion FISH (fluorescence in situ hybridization) is a diagnostic tool used to identify genetic alterations in urinary tract cancers. The assay is capable of detecting chromosomal abnormalities, including those involving chromosomes 3, 7, 17, and 9p21. The sensitivity of the test ranges from approximately 33–74%, with higher performance observed for high-grade disease [7,9].
ImmunoCyt/uCyt+ utilizes the technique of fluorescent immunohistochemistry, with reported sensitivities ranging from 66% to 74% and specificities ranging from 74% to 88% [8,9].
While these initial biomarkers demonstrate superior sensitivity compared to cytology, they have not yet attained the combined sensitivity and Negative Predictive Value (NPV) necessary for cystoscopy replacement [8].
Next-generation urinary biomarkers
A recent development in the field of molecular biology has led to the emergence of a new generation of assays that exhibit significantly enhanced diagnostic capabilities.
The Cxbladder Monitor/Triage/Resolve, a multigene mRNA platform, has been developed that combines five urothelial carcinoma-associated gene transcripts with clinical and phenotypic data. The Cxbladder Monitor exhibited sensitivity ranging from 91% to 93%, accompanied by a negative predictive value (NPV) of 96% to 97% in surveillance populations. This performance surpassed that of cytology (22%), NMP22 ELISA (26%), NMP22 BladderChek (11%), and UroVysion FISH (33%) in direct comparative assessments. In the STREAM randomized controlled trial, which was of landmark significance, Cxbladder Triage achieved 90% sensitivity, 99% negative predictive value, and reduced cystoscopy use by 59% in patients with lower-risk hematuria [7,10,11].
The Xpert Bladder Cancer Monitor (XBCM) is an mRNA-based real-time PCR assay that quantifies the expression levels of five target genes. ABL1, CRH, IGF2, UPK1B, and ANXA10. The overall sensitivity was found to be 84%, while the negative predictive value (NPV) was 93%. It is noteworthy that the sensitivity was observed to be particularly high for low-grade (77%) and pTa (82%) tumors. The DaBlaCa-15 randomized controlled trial demonstrated that alternating XBCM with cystoscopy in high-grade NMIBC reduced cystoscopies from 1,029 to 445 (57% reduction) without affecting recurrence-free survival (risk difference 0.08%, 95% CI −7.2% to 7.4%) [12,13].
The Bladder EpiCheck, a DNA methylation assay, exhibited a pooled sensitivity of 73% (86% for high-grade) and a negative predictive value (NPV) of 90%, thereby demonstrating superior sensitivity compared to cytology while maintaining acceptable specificity [2].
The Uromonitor, the assay under consideration, is a DNA-based assay that is capable of detecting TERT, FGFR3, and KRAS hotspot mutations. A meta-analysis of 1,190 tests demonstrated sensitivity of 80.2%, specificity of 96.9%, and Negative Predictive Value (NPV) of 96.6% [14].
Urine Tumor DNA (utDNA) and Cell-Free DNA are two types of DNA found in urine. The BladMetrix DNA methylation test demonstrated 91% sensitivity for recurrence detection (100% for T1/T2/CIS), with the potential to reduce cystoscopies by 55% and detect 48% of recurrences earlier than cystoscopy. The 2024 guideline amendment from the AUA/SUO specifically highlights urinary cell-free DNA as a technology with “promise for future clinical application” [4,5].
Comparative performance of urinary biomarkers
The following table summarizes the diagnostic performance of key urinary biomarkers in the surveillance setting (Table 1):
| Table 1: Comparative Diagnostic Performance of Conventional and Next-Generation Urinary Biomarkers in NMIBC Surveillance. | |||||||
| Biomarker | Technology | Sensitivity (Overall) | Sensitivity (High-Grade) | Specificity | NPV | FDA Approved | Key Reference |
| Urine Cytology | Morphology | 22–42% | 63–66% | 66–97% | 84–87% | N/A | >[1,2] |
| NMP22 BladderChek | Protein (POC) | 10–11% | 40% | 95% | 86% | Yes | [1,2] |
| BTA stat | Protein (POC) | 60% | 64% | 51% | — | Yes | [1] |
| UroVysion FISH | Chromosomal | 33–74% | — | 74–88% | 92% | Yes | [2,3] |
| ImmunoCyt/uCyt+ | Immunohistochemistry | 66–74% | — | 74–88% | 82–88% | Yes | [3] |
| Cxbladder Monitor | mRNA + clinical data | 91–93% | 95% | — | 96–97% | No* | [2,4] |
| Xpert BC Monitor | mRNA (RT-PCR) | 84% | — | 91% | 93% | No | [5] |
| Bladder EpiCheck | DNA methylation | 73% | 86% | 63% | 90% | No | [6] |
| Uromonitor | DNA mutations | 80% | — | 97% | 97% | No | [7] |
| utDNA (BladMetrix) | DNA methylation | 91% | 100% | — | >99% | No | [8] |
| *Cxbladder is CLIA-certified and commercially available in the US, but is not FDA-cleared as a traditional diagnostic device. This table summarizes the sensitivity, specificity, and negative predictive value (NPV) of key urinary assays. Note the significant improvement in sensitivity and NPV for next-generation mRNA and DNA-based tests compared to traditional urine cytology and early FDA-approved protein markers [2,5-10,12,14]. |
|||||||
Landmark Randomized Controlled Trials
Two randomized controlled trials have provided the first level-1 evidence supporting the use of biomarkers to guide the reduction of cystoscopy procedures.
The DaBlaCa-15 Trial (Xpert Bladder Cancer Monitor) is a research study that is currently underway. This noninferiority Randomized Controlled Trial (RCT) enrolled patients with high-grade non-muscle invasive bladder cancer (NMIBC). The patients were randomized to receive standard cystoscopy at every visit or to undergo alternating X-ray cystography (XBCM) with cystoscopy. The intervention arm underwent a 57% reduction in cystoscopies (445 vs. 1,029), with no statistically significant difference in high-grade recurrence detection (risk difference 0.08%). A notable finding was the observation of an anticipatory positive effect, wherein XBCM successfully detected molecular positivity before the occurrence of visible cystoscopic recurrence [13].
The STREAM Trial (Cxbladder Triage) is a prospective Randomized Controlled Trial (RCT) that was conducted on patients with microhematuria. The study revealed that a marker-based approach using Cxbladder Triage led to a 59% reduction in cystoscopy utilization among lower-risk patients. Furthermore, the approach exhibited a 99% Negative Predictive Value (NPV) for the detection of urothelial carcinoma [11].
A systematic review and network meta-analysis of 122 studies, involving 24,252 patients, was conducted in 2026. The review confirmed that novel urine-based assays exhibited pooled sensitivity rates of up to 90% and specificity rates of up to 92%. These diagnostic values were found to be significantly higher compared to those of cytology [15].
Current guideline positions
Notwithstanding the encouraging data, prevailing guidelines continue to exercise caution:
According to the NCCN (2026), the greatest clinical value of biomarkers lies in their ability to inform the subsequent evaluation of patients with subjective findings at the time of office cystoscopy or in the context of abnormal cytology accompanied by negative imaging. However, the capacity of these biomarkers to supplant the necessity for cystoscopy or cytology remains uncertain and necessitates the implementation of randomized clinical trials to ascertain their efficacy [3].
The AUA/SUO (2024) acknowledges a “limited role for urinary biomarkers to replace cystoscopic surveillance,” but recognizes that advances in the Cxbladder platform and urinary cell-free DNA “hold promise for future clinical application” [4].
For high-risk NMIBC, the NCCN suggests that clinicians may “consider urinary urothelial tumor markers (category 2B)” during year 1 surveillance [3].
The EAU (2024) maintains the most conservative position, designating cystoscopy as the only “strong recommendation” for NMIBC follow-up and explicitly stating there is “no recommendation for routine use of molecular markers to assist in prognosis or to guide therapy.” The EAU does not formally endorse the use of urinary biomarkers for the initial evaluation of hematuria, nor do they advocate for their replacement of cystoscopy in surveillance protocols. However, affiliated reviews acknowledge that “implementation of these tests into daily practice is already occurring,” despite the absence of formal endorsement from official guidelines. The EAU 2024 guidelines employ the 2021 EAU NMIBC progression scoring model for risk stratification and mandate cystoscopy at each scheduled surveillance visit across all risk groups [16-18].
While next-generation assays demonstrate high sensitivity, several limitations hinder their universal adoption. First, the detection of low-grade Ta tumors remains a challenge, with sensitivities often falling below 80%, raising concerns about missed recurrences. Second, most available data stems from observational cohorts rather than long-term randomized trials assessing progression rates. Furthermore, the clinical “anticipatory positive” effect, where a biomarker is positive before a visible lesion appears, requires standardized management protocols to avoid over-treatment or patient anxiety. Finally, the cost-effectiveness of these markers in diverse healthcare systems remains to be fully elucidated through formal economic modeling.
Proposed future algorithm: Risk-stratified biomarker-guided surveillance
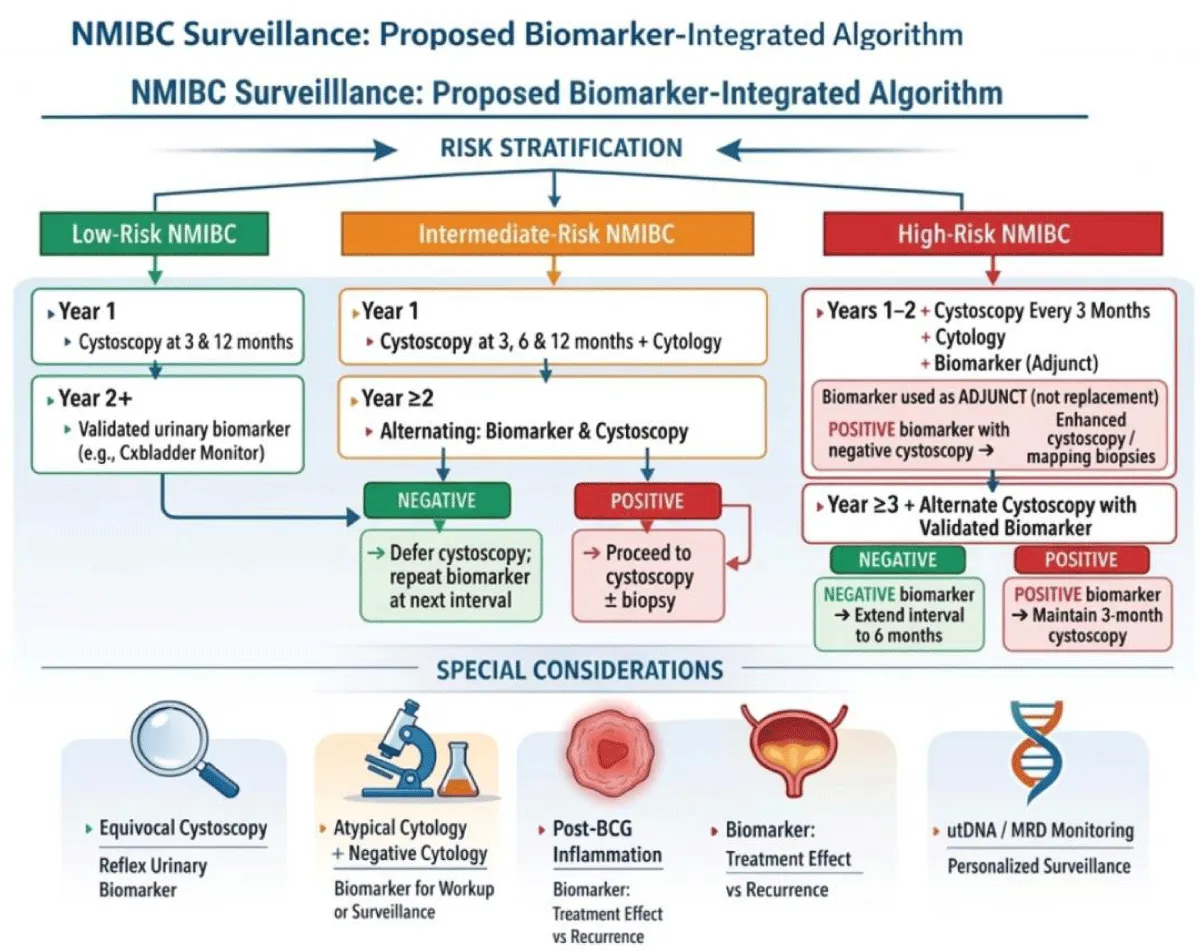
The following algorithm is a theoretical framework predicated on mounting evidence from recent trials for integrating urinary biomarkers into NMIBC surveillance. It must be explicitly stated that this model is hypothetical and has not been endorsed by current NCCN, AUA, or EAU guidelines (Figure 1).
Figure 1: Proposed (Hypothetical) Risk-Stratified, Biomarker-Integrated Algorithm for NMIBC Surveillance. (Delineates a potential future clinical framework for reducing cystoscopy frequency in Low-Risk, Intermediate-Risk, and High-Risk patients using validated urinary biomarkers, and is not intended for current routine clinical use) Created in https://BioRender.com
Barriers to implementation and future directions
However, before this approach can be widely adopted, several challenges must be addressed.
Regulatory and validation gaps have been identified as significant issues. The majority of next-generation assays lack formal FDA approval for surveillance indications. A pressing need exists for large, multicenter, prospective Randomized Controlled Trials (RCTs) that assess long-term oncological outcomes [3,4].
The low-grade tumor detection sensitivity of most biomarkers (44%–86%) remains a significant challenge, particularly with regard to low-grade Ta tumors. The clinical significance of missing small, low-grade recurrences that may be self-limiting remains a subject of debate [2,10].
Standardization and heterogeneity in study populations, reference standards, and cutoff thresholds limit the ability to make cross-study comparisons [15,19].
While biomarker testing is less expensive per test than cystoscopy, the cost-effectiveness of biomarker-guided strategies incorporating confirmatory cystoscopies for positive results requires formal health-economic modeling.
The advent of emerging technologies, such as urine tumor DNA (utDNA) for Minimal Residual Disease (MRD) monitoring, signifies a paradigm shift in the field. This technological advancement holds the promise of not only detecting recurrence but also facilitating personalized surveillance intervals based on the molecular disease burden. The utilization of next-generation sequencing and machine learning-based platforms holds promise for further enhancing diagnostic accuracy and risk stratification [1,5,20].
The prevailing paradigm of bladder cancer surveillance, which is predicated on exclusive reliance on cystoscopy, is undergoing a transition. The initial randomized controlled trials have demonstrated that biomarker-guided strategies can safely reduce cystoscopy frequency by 55–59% without compromising oncological outcomes in selected populations. Next-generation urinary biomarkers, particularly Cxbladder Monitor, Xpert Bladder Cancer Monitor, and urine tumor DNA assays, have achieved the sensitivity and NPV thresholds necessary to serve as reliable “rule-out” tests. Current guidelines appropriately call for prospective Randomized Controlled Trial (RCT) validation before endorsing cystoscopy replacement. However, the trajectory suggests a shift toward a risk-stratified, biomarker-integrated approach that aims to safely reduce cystoscopy frequency rather than replace it entirely. Future protocols should focus on reserving invasive procedures for high-risk clinical scenarios while sparing the majority of patients from unnecessary morbidity, provided that long-term oncological safety is confirmed by ongoing prospective trials.
Declaration
Guarantor of submission: The corresponding author is the guarantor of the submission.
Availability of data and materials: Supporting material is available if further analysis is needed.
Provenance and peer review: Not commissioned, externally peer-reviewed.
- Yang Z, Song F, Zhong J. Urinary biomarkers in bladder cancer: FDA-approved tests and emerging tools for diagnosis and surveillance. Cancers. 2025;17(21):3425. Available from: https://pubmed.ncbi.nlm.nih.gov/41228219/
- Hsia Y, Chiang C, Chiang C, Chiang C, Wang S, Chang Y, et al. Clinical performance of Bladder EpiCheck methylation test for active surveillance of non-muscle invasive bladder cancer: systematic review and meta-analysis. J Clin Oncol. 2023;41(Suppl 16):e16597. Available from: https://ascopubs.org/doi/10.1200/JCO.2023.41.16_suppl.e16597
- National Comprehensive Cancer Network. Bladder cancer. 2026. Available from: https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1417
- Holzbeierlein JM, Bixler BR, Buckley DI, Chang SS, Holmes R, James AC, et al. Diagnosis and treatment of non-muscle invasive bladder cancer: AUA/SUO guideline: 2024 amendment. J Urol. 2024;211(4):533-538. Available from: https://pubmed.ncbi.nlm.nih.gov/38265030/
- Vedeld HM, Pharo H, Sørbø AK, Brandt-Winge S, Five M, Jeanmougin M, et al. Distinct longitudinal patterns of urine tumor DNA in patients undergoing surveillance for bladder cancer. Mol Oncol. 2024;18(11):2684-2695. Available from: https://pubmed.ncbi.nlm.nih.gov/38720532/
- Ecke TH, Meisl CJ, Schlomm T, Rabien A. Performance of urinary markers in patients with suspicious cystoscopy during follow-up of recurrent non-muscle invasive bladder cancer: BTA Stat, NMP22 BladderChek, UBC Rapid Test, CancerCheck UBC Rapid VISUAL, and Uromonitor in comparison to cytology. Urology. 2025;197:119-125. Available from: https://www.researchgate.net/publication/386319602_Performance_of_urinary_markers_in_patients_with_suspicious_cystoscopy_during_follow-up_of_recurrent_non-muscle_invasive_bladder_cancer_BTA_statR_NMP22R_BladderChekR_UBCR_Rapid_Test_CancerCheckR_UBCR_r
- Lotan Y, O’Sullivan P, Raman JD, Shariat SF, Kavalieris L, Frampton C, et al. Clinical comparison of noninvasive urine tests for ruling out recurrent urothelial carcinoma. Urol Oncol. 2017;35(8):531.e15-531.e22. Available from: https://pubmed.ncbi.nlm.nih.gov/28366272/
- Chou R, Gore JL, Buckley D, Fu R, Gustafson K, Griffin JC, et al. Urinary biomarkers for diagnosis of bladder cancer: a systematic review and meta-analysis. Ann Intern Med. 2015;163(12):922-31. Available from: https://pubmed.ncbi.nlm.nih.gov/26501851/
- Todenhöfer T, Hennenlotter J, Esser M, Mohrhardt S, Aufderklamm S, Böttge J, et al. Stepwise application of urine markers to detect tumor recurrence in patients undergoing surveillance for non-muscle-invasive bladder cancer. Dis Markers. 2014;2014:973406. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC4284969/
- Kavalieris L, O'Sullivan P, Frampton C, Guilford P, Darling D, Jacobson E, et al. Performance characteristics of a multigene urine biomarker test for monitoring for recurrent urothelial carcinoma in a multicenter study. J Urol. 2017;197(6):1419-1426. Available from: https://pubmed.ncbi.nlm.nih.gov/27986532/
- Lotan Y, Daneshmand S, Shore N, Black P, Scarpato KR, Patel A, et al. A multicenter prospective randomized controlled trial comparing Cxbladder triage to cystoscopy in patients with microhematuria: the safe testing of risk for asymptomatic microhematuria trial. J Urol. 2024;212(1):41-51. Available from: https://www.auajournals.org/doi/pdf/10.1097/JU.0000000000003991
- Pichler R, Fritz J, Tulchiner G, Klinglmair G, Soleiman A, Horninger W, et al. Increased accuracy of a novel mRNA-based urine test for bladder cancer surveillance. BJU Int. 2018;121(1):29-37. Available from: https://pubmed.ncbi.nlm.nih.gov/28941000/
- Dreyer T, Brandt S, Fabrin K, Azawi N, Vásquez JL, Ernst A, et al. Use of the Xpert bladder cancer monitor urinary biomarker test for guiding cystoscopy in high-grade non-muscle-invasive bladder cancer: results from the randomized controlled DaBlaCa-15 trial. Eur Urol. 2025;88(1):23-30. Available from: https://www.sciencedirect.com/science/article/pii/S0302283825001915
- Kravchuk AP, Wolff I, Gilfrich C, Wirtz RM, Soares P, Braun K, et al. Urine-based biomarker test Uromonitor® in the detection and disease monitoring of non-muscle-invasive bladder cancer - a systematic review and meta-analysis of diagnostic test performance. Cancers. 2024;16(4):753. Available from: https://pubmed.ncbi.nlm.nih.gov/38398144/
- Siguencia F, Heard J, Smith SM, Pagano I, Furuya H, Wu X, et al. Diagnostic performance of urinary biomarker tests in detecting bladder cancer: a systematic review, meta-analysis, and network meta-analysis. Urology. 2026;199:S0090-4295(26)00074-9. Available from: https://pubmed.ncbi.nlm.nih.gov/41651230/
- Gontero P, Birtle A, Capoun O, Compérat E, Dominguez-Escrig JL, Liedberg F, et al. European Association of Urology guidelines on non-muscle-invasive bladder cancer (TaT1 and carcinoma in situ)—a summary of the 2024 guidelines update. Eur Urol. 2024;86(6):531-549. Available from: https://www.sciencedirect.com/science/article/pii/S0302283824025144
- Compérat E, Amin MB, Cathomas R, Choudhury A, De Santis M, Kamat A, et al. Current best practice for bladder cancer: a narrative review of diagnostics and treatments. Lancet. 2022;400(10364):1712-1721. Available from: https://pubmed.ncbi.nlm.nih.gov/36174585/
- Babjuk M, Burger M, Capoun O, Cohen D, Compérat EV, Escrig JLD, et al. European Association of Urology guidelines on non-muscle-invasive bladder cancer (Ta, T1, and carcinoma in situ). Eur Urol. 2022;81(1):75-94. Available from: https://pubmed.ncbi.nlm.nih.gov/34511303/
- Soorojebally Y, Neuzillet Y, Roumiguié M, Lamy PJ, Allory Y, Descotes F, et al. Urinary biomarkers for bladder cancer diagnosis and NMIBC follow-up: a systematic review. World J Urol. 2023;41(2):345-359. Available from: https://pubmed.ncbi.nlm.nih.gov/36592175/
- Guo X, He L, Yang K, Han H, Wei Y, Gong Y, et al. Clinical effectiveness of urine DNA for minimal residual disease (MRD) monitoring of urothelial carcinoma in urine: a multicenter, prospective, observational study. J Clin Oncol. 2025;43(Suppl 16):TPS4617. Available from: https://ascopubs.org/doi/10.1200/JCO.2025.43.16_suppl.TPS4617